Per- and polyfluoroalkyl substances, or PFAS, continue to be a frequent topic of conversation when it comes to their potential impact on the environment and communities. These man-made chemicals, which can be found in food takeout boxes, cleaning products, cookware or even personal hygiene products, have emerged at the forefront of conversation in the water industry.
In April 2024, the U.S. Environmental Protection Agency (EPA) released regulations for PFAS limits in municipal drinking water. This ruling greatly impacts municipalities and the water industry as a whole. The EPA’s regulations include:
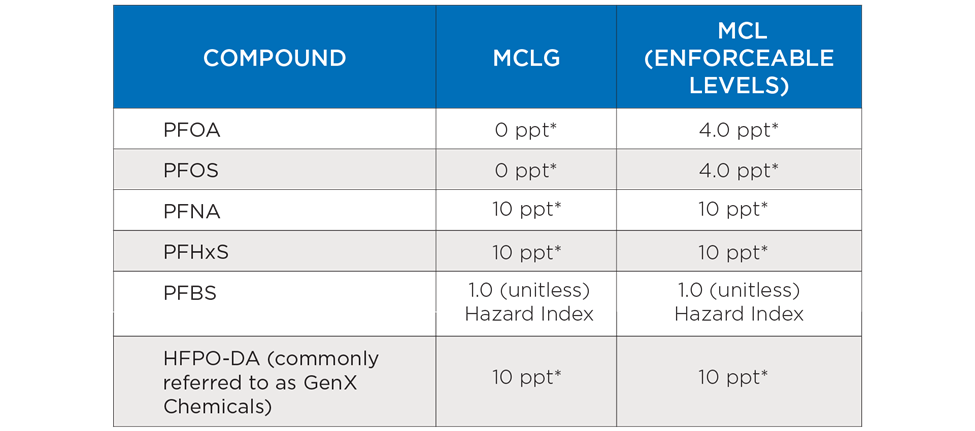
Proposed regulations, which would determine and regulate limits, include:
- Maximum contaminant level (MCL) of 4 parts per trillion for perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), with a MCL goal of 0 parts per trillion
- MCL goal of 10 parts per trillion for perfluorohexanesulfonic acid (PFHxS), perfluorononanoic acid (PFNA) and Gen X (HFPO-DA) chemicals.
- An enforceable limit on a Hazard Index PFNA, PFHxS, perfluorobutane sulfonate (PFBS) and HFPO-DA.
For municipalities and water utilities, these can be uncertain times with changes to treatment and monitoring efforts coming soon. The EPA outlined a five-year compliance schedule for this new regulation with three years to complete initial monitoring and five years to implement solutions.
There are several steps that can be taken to successfully navigate these upcoming regulations:
- Step 1: PFAS Testing
The first step to begin PFAS treatment is testing. This will help determine if PFAS is present within a municipality’s water supply, as well as the breakdown of constituents. - Step 2: Stakeholder Education
Water municipality leadership can host community meetings to help educate local residents and business owners on PFAS and how the chemicals will be treated. - Step 3: Review Facility Infrastructure
It’s important to review existing infrastructure to help identify how a PFAS treatment solution would fit into the system. - Step 4: Consider Pilot Testing
While not currently required, pilot testing can help water municipalities leadership make a more informed decision. The three EPA-approved PFAS treatment methods, granular activated carbon, ion exchange and reverse osmosis, each have differing strengths and weaknesses. Determining the estimated media life or percentage recovery for a water municipality’s specific water source will help determine the optimal treatment technology. Additionally, there are several other PFAS treatment technologies that are not yet approved by the EPA. - Step 5: Review Funding Options
Funding to address emerging contaminants in drinking water is available through the Infrastructure Investment and Jobs Act, which is anticipated to be around $9 billion. These federal funds are distributed through state clean water agencies. - Step 6: Complete Alternative Analysis
After the pilot test data has been received, complete an alternatives analysis of the different options. Evaluating the capital cost and operational and maintenance costs will help water municipality leadership make the optimal long-term decision for the existing system.
There are thousands of PFAS compounds identified and the full health impacts of PFAS are still being realized. Further, PFAS-containing products are still being manufactured and used in many places. However, the EPA has elected to regulate six PFAS compounds in drinking water. It has never been more important to gain a clear understanding of the regulations to come and how to prepare.
Protecting clean and reliable water is crucial to maintaining a healthy community. Learn how to proactively plan for emerging contaminants with an experienced team on your side.

